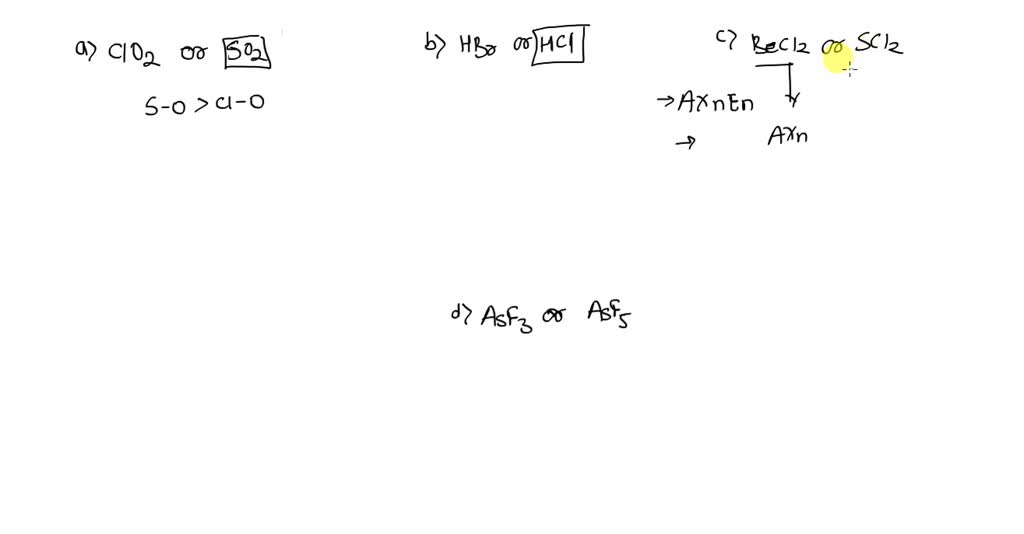
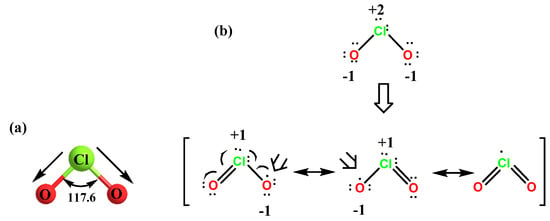
SOLVED:Which molecule in each pair has the greater dipole moment? Give the reason for your choice. (a) ClO2 or SO2 (b) HBr or HCl (c) BeCl2 or SCl2 (d) AsF3 or AsF5

SOLVED: Which molecule in each pair has the greater dipole moment? Give the reason for your choice. (a) ClO2 or SO2 (b) HBr or HCl (c) BeCl2 or SCl2 (d) AsF3 or

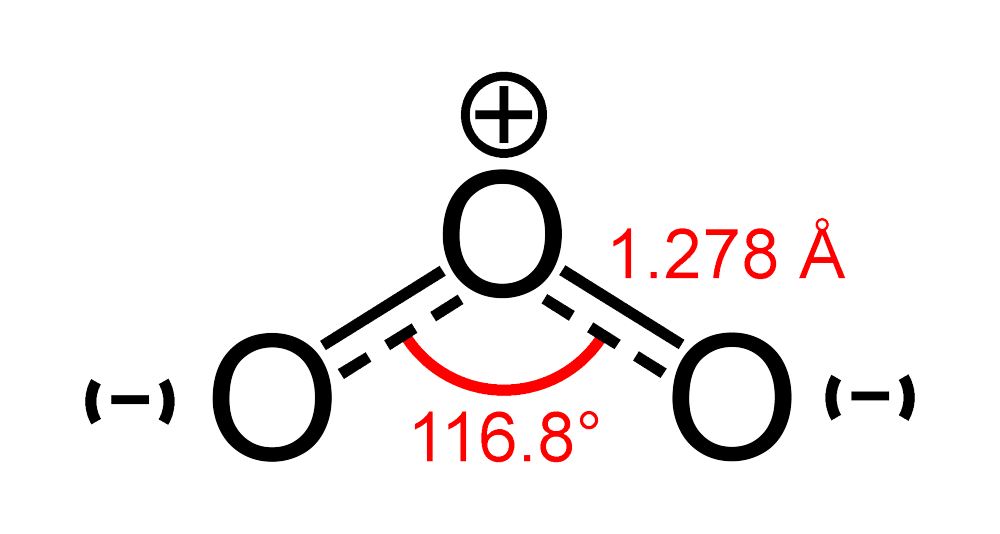
Determine if each compound or ion below has a dipole moment. (a) Carbonate ion (CO_3 ^{2-}) | Homework.Study.com

Popelle The molecule which has zero dipole moment is (1) CH2Cl2 (2) BF3 (3) NF3 (4) CIOZ 171:of the following model best doorbooth
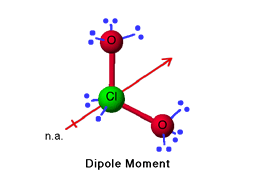
An Integrative Synthetic, Spectroscopic, and Computational Study of the Free Radical Chlorine Dioxide and its Interactions with






![Solved Question No.2 [CLO2] a) Two metal plates as shown in | Chegg.com Solved Question No.2 [CLO2] a) Two metal plates as shown in | Chegg.com](https://media.cheggcdn.com/study/7fe/7fe0c828-7fdc-455e-880f-258701bf58a6/image)